Our Identity and Mission
The Institute of Science for Observation, Standardization, Assessment and Frontier Development (ISOSAF) was established as a high-level scientific entity to address the growing complexity of global industrial demands. In an era where “Quality” is no longer a static goal but a multifaceted scientific discipline, ISOSAF serves as a cornerstone for enterprises in the Pharmaceutical, Cosmetic, and Food sectors.
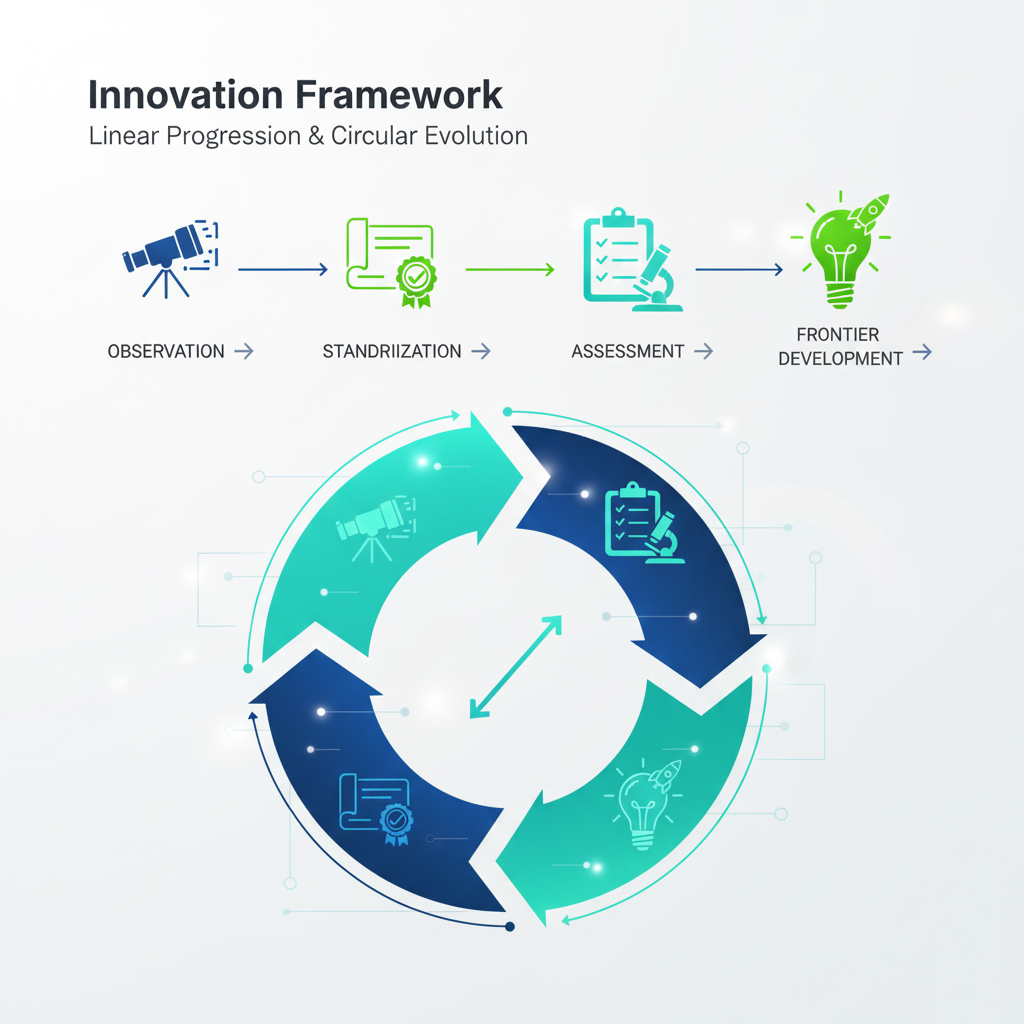
Our name is not merely a title; it is our operational roadmap. Each component of the ISOSAF acronym represents a critical stage in the lifecycle of quality and safety.

The Four Pillars
Observation
At ISOSAF, everything begins with Observation. We believe that you cannot standardize what you do not accurately measure.
Scientific Monitoring: We employ high-precision monitoring techniques to observe industrial environments, manufacturing processes, and biological impacts.
Data-Driven Insights: Our observation phase involves the collection of empirical data, from air quality and chemical runoff in cosmetic plants to the microbial stability of pharmaceutical production lines.
The Goal: To provide a transparent baseline of “current reality” before any certification or improvement begins.
Standardization
Once data is observed, it must be governed. Standardization is the core of our technical expertise.
Framework Creation: We assist organizations in translating complex international regulations (ISO, GMP, Halal) into actionable internal standards.
Beyond Compliance: ISOSAF works with regulatory bodies to develop “Frontier Standards”—benchmarks that anticipate future legal requirements in the EU, USA (FDA), and ASEAN markets.
The Goal: To ensure every product and process follows a repeatable, high-quality, and legally defensible blueprint.
Assessment
Assessment is the bridge between a standard and its realization. It is where our independence as an Institute adds the most value.
Rigorous Auditing: Our experts conduct deep-dive assessments to verify that “Standardization” is being lived in daily operations.
Risk Analysis: We perform toxicological assessments for cosmetics, safety integrity audits for food, and efficacy assessments for pharmaceuticals.
Certification Readiness: Our assessment reports serve as the definitive proof of compliance, providing the necessary documentation for ISO or GMP certification.
The Goal: To provide an objective, third-party validation that builds trust with consumers and global regulators.
Frontier Development
The world moves fast, and standards must evolve. Frontier Development is what sets ISOSAF apart from traditional consulting firms.
Applied Research: We invest in the research of new materials, sustainable chemical processes, and “Green” hazardous waste treatment technologies.
Future-Proofing: We help our partners innovate by integrating “frontier” concepts—such as circular economy principles in pharmaceutical waste or AI-driven quality control.
The Goal: To ensure that our clients are not just meeting today’s standards, but are leaders in the innovations of tomorrow.
Our Commitment to Industry & Law
ISOSAF operates under a strict framework of legal integrity. Our consultants and scientists provide guidance that is cross-referenced with:
International Treaties: Aligning with WHO, FAO, and ISO global guidelines.
National Legislation: Adhering strictly to Environmental Protection Laws, Food Safety Laws, and Pharmaceutical specialized decrees.
Why the Industry Trusts ISOSAF
Multidisciplinary Expertise: Our team consists of PhD-level scientists, environmental engineers, and legal experts in Halal and GMP.
State-of-the-Art Facilities: We leverage advanced laboratory observations to back our certifications with hard science.
Global Vision, Local Context: We understand the nuances of the Vietnamese market while maintaining the rigorous standards expected by international partners in Europe and North America.
"Science is our foundation; standards are our compass. We are committed to delivering absolute transparency and precision in every assessment process."
General Director: Lukas Hoffmann
